Introduction
Steel is – by a long way – the most commonly used metallic material. Indeed, more steel is produced each year than all of the other metals put together. One of the reasons for steel’s ubiquity is the vast range of properties it can be designed to exhibit. One way of changing its properties is by altering the alloying elements; another is by heat treating the steel in a variety of ways, and it is that we will cover in this section.
By the end of the section, you will Have a knowledge and understanding of how heat treatments can change the properties of steels, including the basics of heat treatment, quench hardening, tempering, surface hardening and annealing – both normalising and stress relieving.
Reasons for Heat Treatment of Steels
Heat treatment of steels may be carried out for a number of reasons, and surprisingly it can sometimes be necessary to use heat treatment to return the metal to a previous state, in other words it can be used as an interim process to allow further processing to occur, e.g., softening after cold working which tends to harden metals and render them brittle.
Alternatively, it may be the end process in itself, being used to produce the desired properties in the final product, e.g., hardness or ductility. It can also be used to produce the desired properties in the bulk material. Whatever the intention of the heat treatment, they involve defined heating and cooling regimes that result in changes to the microstructure and the subsequent properties of the metal.
Basic Process
Whatever the intention or type of the process, it will follow the same basic procedure, namely the steel is heated and held at the temperature to ensure all the steel reaches the desired temperature. What the temperature is and how long the steel is held at it will depend on the process.
After heating, the steel is then cooled, the way in which it is cooled will also affect the end properties of the metal. Cooling can be undertaken in the furnace (slow), in air (intermediate), or quenched (rapid). Quenching can take place in water or another substance such as oil, again it depends on the specific intended outcome.

Gareth Bradley, Perth College UHI
Main Heat Treatment Processes
There are a vast array of heat treatment process to which steel can be subjected, but there are certain processes that are much more common than others. The most common of them are outlined in the table below, along with their intention and how they are carried out.
| Quench hardening (to increase hardness and strength) | Heating and rapid cooling |
| Tempering (to increase the toughness after quenching with some loss of hardness and strength) | Heating and intermediate cooling |
| Annealing (to soften and increase ductility - maybe an interim process) | Heating and slow cooling |
| Normalising (to soften and give a uniform microstructure) | |
| Stress relieving (to remove residual stresses and manufacturing operations) | |
| Surface treatments (to harden the surface region) | Heat and the addition of carbon or nitrogen |
Changes to the Microstructure
All heat treatment changes the grain size in the steel and may change the crystal structure depending on the process employed.
The properties of the heat-treated steel depend on:
- The composition of the steel.
- The temperature to which it is heated.
- The rate of cooling.
Once heat treated, the steel will develop an unique microstructure, each of which will have it’s own properties, which will affect the properties of the steel overall. Different heat treatments are used depending on the type of steel and the desired properties and microstructure required. Heat treatments are tailored to promote the formation of a specific microstructure, thereby developing the steel’s properties to meet application requirements.
Quench Hardening
Quench hardening is one of the more common types of heat treatment in which metal – in this case steel – is heated to a temperature somewhere above normal (room temperature for example), and then rapidly cooled. The temperature to which it is heated is usually above the recrystallisation temperature, and is always below the melting temperature. It may be necessary to hold the material at the temperature for a period of time to ensure equalisation across the material – a process known as “soaking”. Once at the right temperature it is then plunged into a cooling medium such as water, rapidly returning it to room temperature. However the process is carried out, it will affect the hardness of the material. Different quenching media will have different effects on the properties of the material. Not all materials can be quench hardened, materials that can be quench hardened are shown in blue in the diagram.

© A. Henderson, UHI.
The temperature required for hardening will depend on the carbon content of the steel:
- Plain carbon steels <0.3% C will not harden by heat treatment.
- The temperature decreases from ~840 °C as C content increases from 0.3% up to 0.83%, where the temperature is ~723 °C.
- Above 0.8% the temperature increases from ~723 °C depending on the carbon content.
Once heated, and soaked if need be, the steel will then need to be quenched. This can take place in a number of different media:

© A. Henderson, UHI.
Different medias have different severities of quenching. Agitating the media while quenching increases the severity of the quench. Too rapid cooling can cause cracking in complex and heavy sections.
Austenite and Martensite
Austenite is the structure of iron and steels at high temperatures (over ~723°C). It has a face centred cubic structure and up to 2% carbon will dissolve in the structure. For significant quench hardening to occur, all the steel must be austenite (and so must be heated to over 723°C throughout). It takes its name from Sir William Chandler Roberts – Austen, a 19th century metallurgist, not the city in Texas as is sometimes believed.

Figure 1 Microstructure of Austenite. [Wikimedia / CC BY-SA 4.0]
Quenching causes the austenite to be partially or totally transformed into martensite. Martensite is only formed by very rapid cooling from the austenitic microstructure.
Martensite is a very hard form of steel, formed as the rapid cooling means that the carbon atoms do not have time to diffuse out of the crystal microstructure in large quantities.

Figure 2 Microstructure of Martensite. [Wikimedia / CC BY-SA 4.0]
Hardenability
Hardenability of steel is literally how able it is to undergo hardening and how effective the process will be in terms of increasing the hardness of the steel from its starting point. The hardenability of the steel is dependent upon it’s chemical composition.
Many, if not most, of the heat treatable steels are alloy steels rather than plain carbon steels. The addition of nickel, chromium and molybdenum increases the hardenability of the steel. The only common alloying element that appears to decrease hardenability is cobalt.
In large components, not all the section of may harden properly due to the mass effect leading to a slow cooling rate. This can lead to a component being left with a soft core, and in extreme cases could prevent hardening taking place altogether.
Tempering
The hardening process will tend to produce a steel that is very hard, but also rather brittle, lacking in toughness. To counteract this, tempering is often undertaken to modify the microstructure, which gives a tougher, less brittle steel which still retains high strength.
During the tempering process, the metal is heated to a high temperature – though below the melting point – and then cooling it slowly, usually in air. This processes lessens brittleness and reduces internal stresses, which has the effect of toughening the steel.
Appropriate tempering temperatures, the colour of the metal after tempering and example uses are shown in the table.

Gareth Bradley, Perth College UHI
Annealing
Hardening and tempering can create a steel that is both hard and tough, but this can be a problem in itself as it tends to create a material that is very difficult to work with.
For this reason, steels – and many other metals – are often annealed, which softens them and improves their machinability and/or manufacturability.
It can also be used to relive residual (internal) stresses in the material that may have been caused by previous manufacturing processes such as rolling or forging.
Different annealing processes are used depending on the required treatment or outcome, but they all involve heating the material above recrystallisation temperature, holding it at an appropriate temperature for the appropriate length of time, and then cooling the material:
- Heat up followed by slow cooling.
- Recovery.
- Recrystallisation.
- Grain growth.

Figure 3 The annealing process - © Gareth Bradley (UHI).
Annealing Types
There are four major types of annealing, as represented in the diagram:
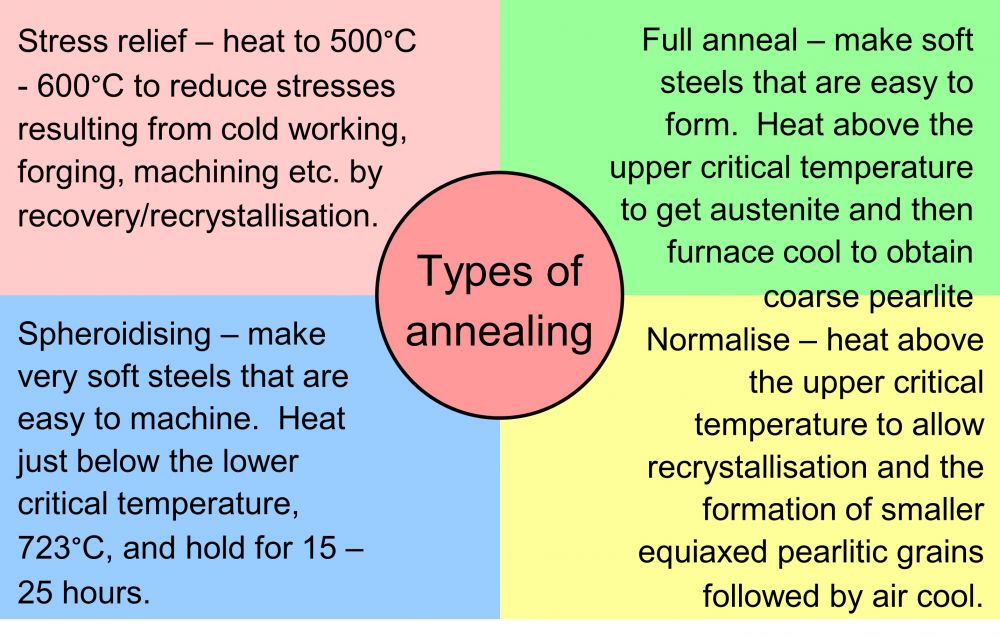
Figure 4 The Four major types of annealing
Surface Hardening
Surface hardening – or case hardening as it is more commonly known – are treatments that are employed to harden the surface region of a component to improve its wear resistance, but without reducing the toughness of the inner.
A gear wheel is the classic example – a gear wheel needs hard teeth to prevent wear, but a tough inner to resist fracturing.
There are two main types of case hardening – carburising and nitriding.

Figure 5 Gear teeth. [Pixabay]
Carburising
During Carburising carbon is absorbed into the surface region of a low carbon or low alloy steel through heating in the presence of a carbon rich media. Medium and high carbon steels cannot be carburised, as there is already a high level of carbon in the surface area.
The media can be a carbon rich gas such as carbon monoxide (CO), methane (CH4) or propane, (C3H8); a solid such as charcoal, or a liquid such as a bath of molten salt containing sodium cyanide together with sodium carbonate and sodium chloride.
After carburising has taken place, the material can then be subject to quenching and/or tempering to further improve the hardness and toughness.
The image shows a section of carburised steel, and the difference between the surface area and the internal of the object can easily be seen. The thickness of this surface area – known as the case – is determined by the length of time that it spends in contact with the carbon – rich media.

Figure 6 Section of Carburized Steel. [Wikimedia]
Nitriding
The principle of nitriding is similar to that of carburising, except that rather than carbon being absorbed onto the surface of the object, nitrogen is the hardening element.
Nitriding results in a very hard, but also very shallow surface layer. It is created by exposing steel to ammonia gas (NH3) at an elevated temperature in the region of 500°C.
Nitriding cannot be carried out on all types of steel, but rather it is only suitable for certain alloy steels (known as nitriding steels) that contain aluminium (Al), chromium (Cr), molybdenum (Mo), vanadium (V) and/or titanium (Ti), as these react with the nitrogen to form nitrides.
The process is relatively slow, requiring exposure times of 40 – 100 hours, but it does result in an end product that is very hard, meaning that quenching is not required after the treatment, parts are therefore less likely to distort.
For a variety of reasons – including the time taken – it tends to be used for more expensive components. As with carburising, the thickness of the case is determined by the length of time that the component stays in contact with the nitrogen rich-media.
The image shows a particularly advanced form of nitriding, namely pulse plasma ion nitriding. This process is extremely controllable, but the end goal is the same – hardening of the surface.

Figure 7 Nitriding in Canada - Advanced Nitriding Solutions. [Wikimedia / CC BY-SA 4.0]














